Your selection
Research / 24.02.2026
Similar kinases play distinct roles in the brain
Researchers at the Max Delbrück Center show that two closely related enzymes, MNK1 and MNK2, act on the brain to regulate different behaviors. Their findings, published in “Molecular Psychiatry,” could help guide development of more precise therapies for neurological disorders.
Structurally, they look similar: MNK1 and MNK2 belong to the same enzyme family and are best known for regulating how cells make proteins. Their starring role in such a crucial cellular function has cast them into the spotlight as potential drug targets to treat nervous system disorders and chronic pain. But would it matter whether a drug targets only one of them?
In a study published in “Molecular Psychiatry,” researchers led by Rosalba Olga Proce, a doctoral student in the Molecular and Cellular Basis of Behavior lab led by Dr. Hanna Hörnberg at the Max Delbrück Center, set out to determine whether the two enzymes – also called kinases – perform distinct functions in the brain. The team found clear differences. Mice lacking MNK1 showed less interest in newly introduced objects than controls and impaired memory of objects. By contrast, mice without MNK2 appeared normal in object recognition tests but showed enhanced interest in social contacts.
“The behavioral differences we observed suggest that each kinase has a specialized function,” says Proce. “It might be preferable to target each kinase individually when designing drugs.”
Mouse behavior and molecular data
To uncover how these behavioral changes arose, the researchers used genetically engineered mice in which either the MNK1 or MNK2 gene was knocked out. They then combined behavioral testing with large-scale molecular analyses of brain tissue.
The team focused in particular on the cerebral cortex and on synapses – contact points where neurons communicate. Using proteomics, transcriptomics, and phosphoproteomics, they compared protein levels, gene expression, and protein phosphorylation patterns between the two types of mice.
Mice lacking MNK1 had an abundance of ribosomal proteins, which play a central role in transcribing messenger RNA into proteins. By contrast, in mice lacking MNK2, the expression and phosphorylation of proteins responsible for transmitting messages in synapses was reduced.
Notably, the researchers found that the molecular differences between the two kinases were far more pronounced in synapses than in whole-brain samples.
“We were surprised to see these differences,” says Hörnberg. “This suggest that the kinases may have different functions in the cell body compared to synapses.”
Toward more selective therapies
Instead of broadly inhibiting both kinases, future therapies might selectively target one kinase or the other to achieve more specific effects, Hörnberg adds.
She and her colleagues now aim to identify other molecules with which MNK1 and MNK2 interact. They also plan to study in greater detail, how these kinases regulate translation of messenger RNA into proteins during synaptic plasticity – the strengthening or weakening of connections between neurons over time. Synaptic plasticity is the fundamental mechanism underlying learning and memory – enabling the brain to rewire itself based on experience.
By clarifying how protein synthesis is fine-tuned at synapses, the team hopes to lay the groundwork for more precise interventions in neurological and psychiatric disorders.
Text: Gunjan Sinha
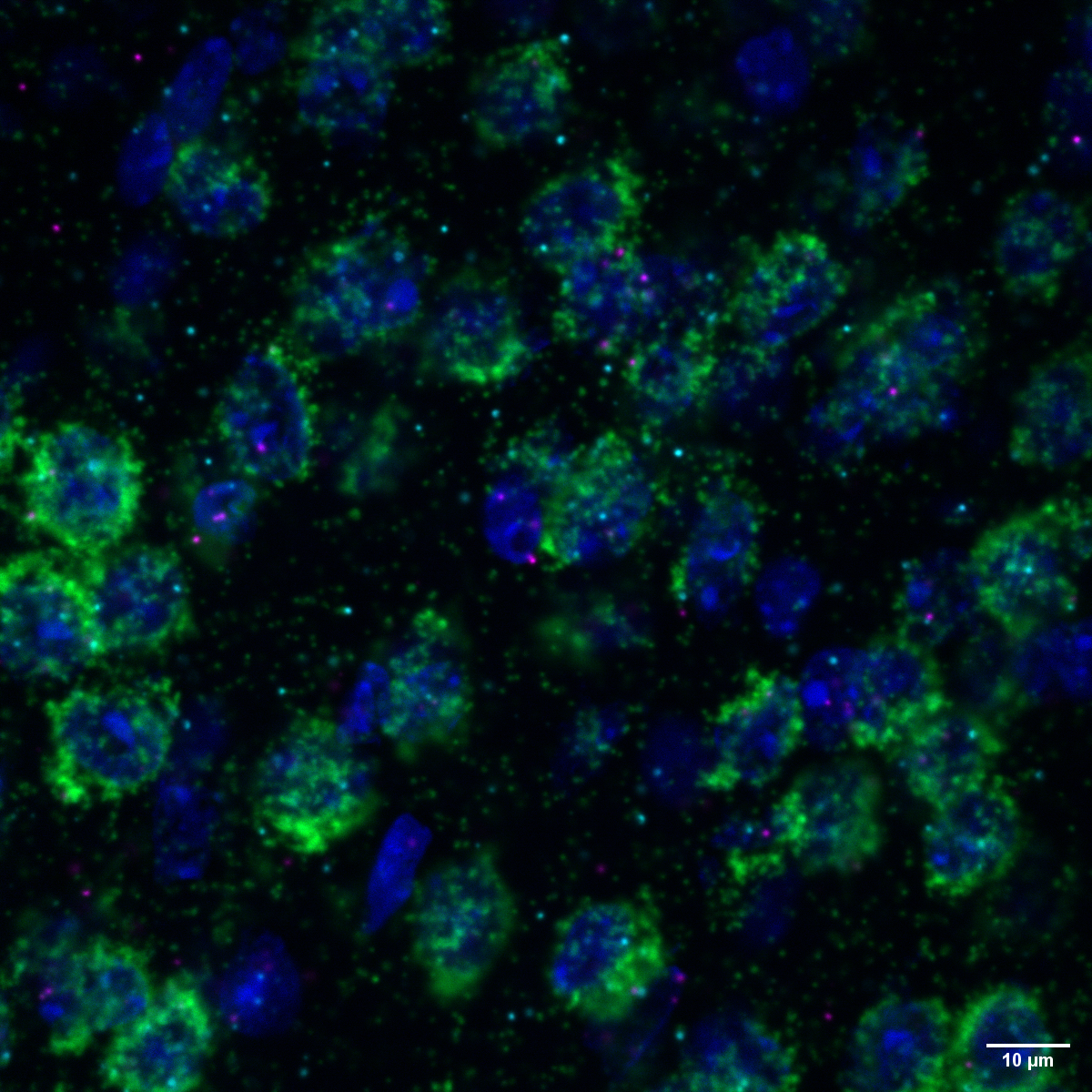
Photo: Section of mouse brain tissue showing that messenger RNA for MNK1 (magenta) and MNK2 kinase (turquoise) – important enzymes that regulate the production of proteins – are both expressed in excitatory neurons (green) in the cortex.
© Erika Uddström, Hörnberg Lab, Max Delbrück Center
Source: Press Release Max Delbrück Center
Similar kinases play distinct roles in the brain
Overview News
News Buch Berlin
Equality in research drives innovation
Marking International Women's Day, Federal Research Minister Dorothee Bär met with female researchers at the Max Delbrück Center
more ...T-knife Therapeutics Announces Authorization of Clinical Trial Application for TK-6302, A Multi-Armored, CRISPR based T cell Therapy for Solid Tumors
Phase 1 ATLAS trial to evaluate TK-6302 in patients with advanced PRAME-positive solid tumors authorized for initiation
more ...Gut microbes affect the heart via the brain
Researchers at the Max Delbrück Center have uncovered how a bacterial metabolite produced in the gut regulates heart function through specific brain cells. The findings, published in “Circulation Rese...
more ...Events Buch Berlin
13.03.2026, 16:00
Silent Disco im Bürgerhaus
für Jung und Alt
more ...17.03.2026, 18:00
Campus-Kino: "Greenpeace - wie alles begann"
Der Freundeskreis des Max Delbrück Center lädt herzlich ein!
more ...18.03.2026, 08:30
vocatium Berlin focus 2026: Fachmesse für Ausbildung+Studium
Entdecke berufliche Möglichkeiten, informiere dich und sprich mit Expert:innen über Einstieg, Voraussetzungen und Bewerbung.
more ...






